Principle Of Size Exclusion Chromatography
 Equipment for running size-exclusion chromatography. The buffer is pumped through the cavalcade (right) by a figurer-controlled device. | |
| Acronym | SEC |
|---|---|
| Classification | Chromatography |
| Analytes | macromolecules constructed polymers biomolecules |
| Manufacturers | Cytiva, Bio-Rad, Bio-Works, emp Biotech, Knauer. |
| Other techniques | |
| Related | High-performance liquid chromatography Aqueous normal-phase chromatography Ion commutation chromatography Micellar liquid chromatography |
Size-exclusion chromatography (SEC), also known as molecular sieve chromatography,[1] is a chromatographic method in which molecules in solution are separated by their size, and in some cases molecular weight.[2] It is usually practical to big molecules or macromolecular complexes such every bit proteins and industrial polymers. Typically, when an aqueous solution is used to transport the sample through the column, the technique is known every bit gel-filtration chromatography, versus the name gel permeation chromatography, which is used when an organic solvent is used as a mobile phase. The chromatography column is packed with fine, porous beads which are commonly composed of dextran, agarose, or polyacrylamide polymers. The pore sizes of these beads are used to estimate the dimensions of macromolecules.[1] SEC is a widely used polymer characterization method because of its ability to provide proficient molar mass distribution (Mw) results for polymers.
Applications [edit]
The principal application of gel-filtration chromatography is the fractionation of proteins and other water-soluble polymers, while gel permeation chromatography is used to clarify the molecular weight distribution of organic-soluble polymers. Either technique should not be dislocated with gel electrophoresis, where an electric field is used to "pull" or "push" molecules through the gel depending on their electrical charges. The corporeality of time a solute remains within a pore is dependent on the size of the pore. Larger solutes will have access to a smaller volume and vice versa. Therefore, a smaller solute volition remain inside the pore for a longer flow of fourth dimension compared to a larger solute.[3]
Some other utilize of size exclusion chromatography is to examine the stability and characteristics of natural organic matter in water.[4] In this method, Margit B. Muller, Daniel Schmitt, and Fritz H. Frimmel tested water sources from dissimilar places in the globe to determine how stable the natural organic matter is over a period of fourth dimension.[4] Even though, size exclusion chromatography is widely utilized to study natural organic fabric, there are limitations. One of these limitations include that in that location is no standard molecular weight marking;[iv] thus, at that place is nothing to compare the results back to. If precise molecular weight is required, other methods should exist used.
Advantages [edit]
The advantages of this method include practiced separation of large molecules from the pocket-sized molecules with a minimal volume of eluate,[5] and that various solutions can be applied without interfering with the filtration process, all while preserving the biological activity of the particles to separate. The technique is generally combined with others that farther dissever molecules by other characteristics, such every bit acidity, basicity, charge, and affinity for certain compounds. With size exclusion chromatography, there are short and well-defined separation times and narrow bands, which pb to good sensitivity. In that location is also no sample loss because solutes practise non collaborate with the stationary phase.
The other advantage to this experimental method is that in certain cases, it is feasible to decide the approximate molecular weight of a compound. The shape and size of the chemical compound (eluent) determine how the compound interacts with the gel (stationary stage). To determine guess molecular weight, the elution volumes of compounds with their corresponding molecular weights are obtained and then a plot of "Thouav" vs "log(Mw)" is fabricated, where and Mw is the molecular mass. This plot acts every bit a scale curve, which is used to approximate the desired compound's molecular weight. The Ve component represents the book at which the intermediate molecules elute such every bit molecules that have partial access to the beads of the column. In improver, Vt is the sum of the full book between the chaplet and the book within the chaplet. The Vo component represents the book at which the larger molecules elute, which elute in the start.[half dozen] [7] Disadvantages are, for example, that only a limited number of bands can be accommodated because the time scale of the chromatogram is short, and, in general, at that place must be a ten% difference in molecular mass to have a good resolution.[5]
Discovery [edit]
The technique was invented in 1955 by Grant Henry Lathe and Colin R Ruthven, working at Queen Charlotte's Hospital, London.[8] [9] They later on received the John Scott Award for this invention.[x] While Lathe and Ruthven used starch gels every bit the matrix, Jerker Porath and Per Flodin later introduced dextran gels;[xi] other gels with size fractionation properties include agarose and polyacrylamide. A short review of these developments has appeared.[12]
There were also attempts to fractionate synthetic high polymers; withal, it was non until 1964, when J. C. Moore of the Dow Chemical Visitor published his work on the training of gel permeation chromatography (GPC) columns based on cross-linked polystyrene with controlled pore size,[13] that a rapid increase of research action in this field began. It was recognized almost immediately that with proper scale, GPC was capable to provide tooth mass and tooth mass distribution information for synthetic polymers. Because the latter information was hard to obtain by other methods, GPC came rapidly into extensive apply.[fourteen]
Theory and method [edit]

Agarose-based SEC columns used for protein purification on an AKTA FPLC machine
SEC is used primarily for the assay of large molecules such as proteins or polymers. SEC works by trapping smaller molecules in the pores of the adsorbent ("stationary phase"). This process is usually performed within a column, which typically consists of a hollow tube tightly packed with micron-calibration polymer chaplet containing pores of different sizes. These pores may be depressions on the surface or channels through the dewdrop. As the solution travels down the column some particles enter into the pores. Larger particles cannot enter into as many pores. The larger the particles, the faster the elution. The larger molecules simply pass by the pores considering those molecules are too large to enter the pores. Larger molecules therefore flow through the column more apace than smaller molecules, that is, the smaller the molecule, the longer the retention time.
One requirement for SEC is that the analyte does non interact with the surface of the stationary phases, with differences in elution fourth dimension between analytes ideally beingness based solely on the solute volume the analytes tin enter, rather than chemical or electrostatic interactions with the stationary phases. Thus, a small-scale molecule that tin penetrate every region of the stationary phase pore system can enter a full volume equal to the sum of the unabridged pore book and the interparticle volume. This small molecule elutes late (after the molecule has penetrated all of the pore- and interparticle book—approximately 80% of the column volume). At the other extreme, a very large molecule that cannot penetrate whatsoever the smaller pores tin can enter just the interparticle volume (~35% of the column book) and elutes before when this volume of mobile phase has passed through the cavalcade. The underlying principle of SEC is that particles of different sizes elute (filter) through a stationary phase at unlike rates. This results in the separation of a solution of particles based on size. Provided that all the particles are loaded simultaneously or near-simultaneously, particles of the same size should elute together.
However, every bit in that location are various measures of the size of a macromolecule (for instance, the radius of gyration and the hydrodynamic radius), a fundamental problem in the theory of SEC has been the choice of a proper molecular size parameter by which molecules of unlike kinds are separated. Experimentally, Benoit and co-workers constitute an excellent correlation between elution volume and a dynamically based molecular size, the hydrodynamic volume, for several dissimilar concatenation architecture and chemic compositions.[xv] The observed correlation based on the hydrodynamic volume became accustomed as the basis of universal SEC calibration.
Still, the use of the hydrodynamic volume, a size based on dynamical properties, in the interpretation of SEC data is non fully understood.[16] This is because SEC is typically run under low menstruum rate conditions where hydrodynamic factor should have footling effect on the separation. In fact, both theory and calculator simulations assume a thermodynamic separation principle: the separation process is adamant by the equilibrium distribution (partitioning) of solute macromolecules between two phases: a dilute majority solution phase located at the interstitial space and confined solution phases within the pores of cavalcade packing material. Based on this theory, information technology has been shown that the relevant size parameter to the division of polymers in pores is the mean span dimension (mean maximal project onto a line).[17] Although this issue has not been fully resolved, it is likely that the mean span dimension and the hydrodynamic volume are strongly correlated.

Each size exclusion cavalcade has a range of molecular weights that can be separated. The exclusion limit defines the molecular weight at the upper end of the column 'working' range and is where molecules are also big to get trapped in the stationary phase. The lower finish of the range is defined past the permeation limit, which defines the molecular weight of a molecule that is small enough to penetrate all pores of the stationary phase. All molecules below this molecular mass are so small that they elute as a single band.[v]
The filtered solution that is collected at the terminate is known every bit the eluate. The void volume includes whatsoever particles too large to enter the medium, and the solvent volume is known as the cavalcade volume.
Following are the materials which are unremarkably used for porous gel beads in size exclusion chromatography [18]
| Sr. No | Cloth And Trade name | Fractionation range (Molecular mass in Da) |
|---|---|---|
| 1 | Sephadex K-10 | 0 to 700 |
| 2 | Sephadex G-25 | m to 5000 |
| 3 | Sephadex G-50 | 1500 to 30000 |
| four | Sephadex G-75 | 3000 to 70000 |
| 5 | Sephadex K-100 | 4000 to 150000 |
| six | Sephadex G-150 | 5000 to 300000 |
| vii | Sephadex G-200 | 5000 to 800000 |
| eight | Bio-gel P-two | 100 to 1800 |
| 9 | Bio-gel P-6 | grand to 6000 |
| 10 | Bio-gel P-lx | 3000 to 60000 |
| 11 | Bio-gel P-150 | 15000 to 150000 |
| 12 | Bio-gel P-300 | 16000 to 400000 |
| thirteen | Sepharose 2B | 2 10 106 to 25 x 106 |
| xiv | Sepharose 4B | iii x 105 to 3 x 106 |
| fifteen | Sepharose 6B | ten4 to 20 ten 106 |
Factors affecting filtration [edit]

A cartoon illustrating the theory behind size exclusion chromatography
In real-life situations, particles in solution exercise not have a fixed size, resulting in the probability that a particle that would otherwise be hampered by a pore passing right past it. Besides, the stationary-stage particles are not ideally defined; both particles and pores may vary in size. Elution curves, therefore, resemble Gaussian distributions. The stationary phase may also interact in undesirable means with a particle and influence retentiveness times, though corking care is taken by column manufacturers to utilize stationary phases that are inert and minimize this issue.
Like other forms of chromatography, increasing the cavalcade length enhances resolution, and increasing the column diameter increases column capacity. Proper cavalcade packing is of import for maximum resolution: An over-packed column tin collapse the pores in the chaplet, resulting in a loss of resolution. An under-packed column can reduce the relative area of the stationary phase accessible to smaller species, resulting in those species spending less fourth dimension trapped in pores. Unlike affinity chromatography techniques, a solvent head at the top of the column can drastically diminish resolution as the sample diffuses prior to loading, broadening the downstream elution.
Analysis [edit]
In elementary manual columns, the eluent is nerveless in abiding volumes, known as fractions. The more than similar the particles are in size the more likely they are in the same fraction and not detected separately. More advanced columns overcome this problem by constantly monitoring the eluent.
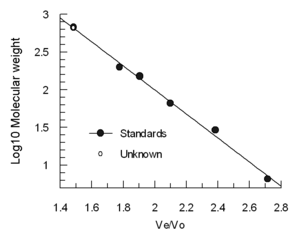
Standardization of a size exclusion cavalcade
The collected fractions are often examined by spectroscopic techniques to determine the concentration of the particles eluted. Common spectroscopy detection techniques are refractive index (RI) and ultraviolet (UV). When eluting spectroscopically similar species (such as during biological purification), other techniques may exist necessary to place the contents of each fraction. It is also possible to analyze the eluent menses continuously with RI, LALLS, Multi-Angle Light amplification by stimulated emission of radiation Low-cal Scattering MALS, UV, and/or viscosity measurements.

SEC chromatogram of a biological sample
The elution volume (Ve) decreases roughly linear with the logarithm of the molecular hydrodynamic volume. Columns are oft calibrated using 4-5 standard samples (e.g., folded proteins of known molecular weight), and a sample containing a very large molecule such as thyroglobulin to make up one's mind the void book. (Blue dextran is non recommended for Vo determination considering information technology is heterogeneous and may give variable results) The elution volumes of the standards are divided past the elution book of the thyroglobulin (Ve/Vo) and plotted confronting the log of the standards' molecular weights.
Applications [edit]
Biochemical applications [edit]
In general, SEC is considered a low-resolution chromatography as information technology does not discern similar species very well, and is therefore often reserved for the final stride of a purification. The technique can decide the fourth construction of purified proteins that have slow substitution times, since information technology tin can be carried out nether native solution weather, preserving macromolecular interactions. SEC tin too assay poly peptide 3rd structure, as information technology measures the hydrodynamic volume (not molecular weight), allowing folded and unfolded versions of the aforementioned poly peptide to exist distinguished. For case, the apparent hydrodynamic radius of a typical protein domain might be xiv Å and 36 Å for the folded and unfolded forms, respectively. SEC allows the separation of these two forms, as the folded course elutes much later due to its smaller size.
Polymer synthesis [edit]
SEC can be used every bit a measure out of both the size and the polydispersity of a synthesized polymer, that is, the ability to observe the distribution of the sizes of polymer molecules. If standards of a known size are run previously, and then a calibration curve can be created to make up one's mind the sizes of polymer molecules of involvement in the solvent called for assay (often THF). In culling way, techniques such equally light scattering and/or viscometry can be used online with SEC to yield absolute molecular weights that practice not rely on calibration with standards of known molecular weight. Due to the difference in size of two polymers with identical molecular weights, the absolute determination methods are, in general, more desirable. A typical SEC organisation tin can quickly (in about half an hr) requite polymer chemists information on the size and polydispersity of the sample. The preparative SEC can be used for polymer fractionation on an analytical scale.
Drawbacks [edit]
In SEC, mass is not measured so much every bit the hydrodynamic volume of the polymer molecules, that is, how much space a item polymer molecule takes up when it is in solution. However, the approximate molecular weight tin can exist calculated from SEC data considering the exact human relationship between molecular weight and hydrodynamic volume for polystyrene can be found. For this, polystyrene is used as a standard. But the relationship between hydrodynamic volume and molecular weight is not the same for all polymers, and so only an judge measurement can exist obtained.[xix] Another drawback is the possibility of interaction between the stationary phase and the analyte. Any interaction leads to a later elution time and thus mimics a smaller analyte size.
When performing this method, the bands of the eluting molecules may be broadened. This tin occur by turbulence caused past the flow of the mobile phase molecules passing through the molecules of the stationary stage. In addition, molecular thermal diffusion and friction between the molecules of the glass walls and the molecules of the eluent contribute to the broadening of the bands. Besides broadening, the bands likewise overlap with each other. As a issue, the eluent usually gets considerably diluted. A few precautions tin be taken to forbid the likelihood of the bands broadening. For instance, one tin utilize the sample in a narrow, highly full-bodied band on the top of the cavalcade. The more concentrated the eluent is, the more than efficient the procedure would be. However, it is not always possible to concentrate the eluent, which can exist considered equally one more disadvantage.[7]
Absolute size-exclusion chromatography [edit]
Absolute size-exclusion chromatography (ASEC) is a technique that couples a light handful instrument, most commonly multi-angle light scattering (MALS) or another form of static light handful (SLS), but possibly a dynamic light scattering (DLS) instrument, to a size-exclusion chromatography system for absolute molar mass and/or size measurements of proteins and macromolecules as they elute from the chromatography system.
The definition of "accented" in this example is that calibration of retention time on the column with a gear up of reference standards is not required to obtain tooth mass or the hydrodynamic size, often referred to as hydrodynamic diameter (DH in units of nm). Non-ideal cavalcade interactions, such equally electrostatic or hydrophobic surface interactions that attune memory time relative to standards, do not impact the concluding result. Likewise, differences between conformation of the analyte and the standard have no outcome on an absolute measurement; for instance, with MALS analysis, the molar mass of inherently disordered proteins are characterized accurately even though they elute at much earlier times than globular proteins with the same molar mass, and the aforementioned is true of branched polymers which elute tardily compared to linear reference standards with the same molar mass.[20] [21] [22] Some other do good of ASEC is that the molar mass and/or size is determined at each signal in an eluting top, and therefore indicates homogeneity or polydispersity within the meridian. For example, SEC-MALS analysis of a monodisperse protein will prove that the entire peak consists of molecules with the same molar mass, something that is not possible with standard SEC analysis.
Determination of molar mass with SLS requires combining the light scattering measurements with concentration measurements. Therefore SEC-MALS typically includes the calorie-free scattering detector and either a differential refractometer or UV/Vis absorbance detector. In addition, MALS determines the rms radius R1000 of molecules above a certain size limit, typically ten nm. SEC-MALS tin therefore clarify the conformation of polymers via the relationship of molar mass to R1000. For smaller molecules, either DLS or, more ordinarily, a differential viscometer is added to determine hydrodynamic radius and evaluate molecular conformation in the same way.
In SEC-DLS, the sizes of the macromolecules are measured equally they elute into the menstruum jail cell of the DLS instrument from the size exclusion column set. The hydrodynamic size of the molecules or particles are measured and not their molecular weights. For proteins a Marking-Houwink type of calculation can be used to estimate the molecular weight from the hydrodynamic size.
A major advantage of DLS coupled with SEC is the ability to obtain enhanced DLS resolution.[23] Batch DLS is quick and unproblematic and provides a directly measure of the boilerplate size, but the baseline resolution of DLS is a ratio of 3:1 in diameter. Using SEC, the proteins and protein oligomers are separated, allowing oligomeric resolution. Assemblage studies can likewise be washed using ASEC. Though the aggregate concentration may not be calculated with light scattering (an online concentration detector such every bit that used in SEC-MALS for tooth mass measurement also determines aggregate concentration), the size of the aggregate tin be measured, only express past the maximum size eluting from the SEC columns.
Limitations of ASEC with DLS detection include menses-rate, concentration, and precision. Considering a correlation function requires anywhere from 3–vii seconds to properly build, a express number of data points tin can exist collected across the summit. ASEC with SLS detection is non limited by catamenia rate and measurement fourth dimension is essentially instantaneous, and the range of concentration is several orders of magnitude larger than for DLS. However, molar mass assay with SEC-MALS does require accurate concentration measurements. MALS and DLS detectors are ofttimes combined in a single musical instrument for more than comprehensive absolute analysis post-obit separation by SEC.
See too [edit]
- PEGylation
- Gel permeation chromatography
References [edit]
- ^ a b Garrett RH, Grisham CM (2013). Biochemistry (5th ed.). Belmont, CA: Brooks/Cole, Cengage Learning. p. 108. ISBN9781133106296. OCLC 1066452448.
- ^ Paul-Dauphin, Due south; Karaca, F; Morgan, TJ; et al. (six Oct 2007). "Probing Size Exclusion Mechanisms of Complex Hydrocarbon Mixtures: The Effect of Altering Eluent Compositions". Free energy & Fuels. 6. 21 (6): 3484–3489. doi:10.1021/ef700410e.
- ^ Brooks DE, Haynes CA, Hritcu D, et al. (June 2000). "Size exclusion chromatography does not require pores". Proceedings of the National University of Sciences of the United States of America. 97 (13): 7064–7. Bibcode:2000PNAS...97.7064B. doi:10.1073/pnas.120129097. JSTOR 122767. PMC16499. PMID 10852951.
- ^ a b c Müller MB, Schmitt D, Frimmel FH (i Dec 2000). "Fractionation of Natural Organic Matter by Size Exclusion Chromatography−Properties and Stability of Fractions". Environ Sci Technol. 34 (23): 4867–4872. Bibcode:2000EnST...34.4867M. doi:ten.1021/es000076v.
- ^ a b c Skoog DA, Holler FJ, Crouch SR (2006). "Ch. 28. Liquid Chromatography" (PDF). Principles of instrumental assay (sixth ed.). Belmont, CA: Thomson Brooks/Cole. p. 816. ISBN9780495012016. LCCN 2006926952. OCLC 77224390.
- ^ Rouessac A, Rouessac F (2000). Chemic analysis: modernistic instrumental methods and techniques (Engl. ed.). Chichester: Wiley. pp. 101–103. ISBN978-0471972617. OCLC 635171657.
- ^ a b Ballou DP, Benore M, Ninfa AJ (2008). Fundamental laboratory approaches for biochemistry and biotechnology (2nd ed.). Hoboken, Northward.J.: Wiley. pp. 127–129. ISBN9780470087664.
- ^ Lathe GH, Ruthven CR (August 1955). "The separation of substances on the footing of their molecular weights, using columns of starch and h2o". The Biochemical Journal. 60 (4): xxxiv. PMC1216175. PMID 13249976.
- ^ Lathe GH, Ruthven CR (April 1956). "The separation of substances and estimation of their relative molecular sizes past the employ of columns of starch in water". The Biochemical Journal. 62 (iv): 665–74. doi:x.1042/bj0620665. PMC1215979. PMID 13315231.
- ^ "The John Scott Award Recipients From 1822 - present". garfield.library.upenn.edu . Retrieved iii Jan 2019.
- ^ Porath J, Flodin P (June 1959). "Gel filtration: a method for desalting and grouping separation". Nature. 183 (4676): 1657–9. Bibcode:1959Natur.183.1657P. doi:10.1038/1831657a0. PMID 13666849. S2CID 32287460.
- ^ Eisenstein M (2006). "Adventures in the matrix". Nature Methods. iii (5): 410. doi:ten.1038/nmeth0506-410. ISSN 1548-7105. S2CID 37935968.
- ^ Moore JC (1964). "Gel permeation chromatography. I. A new method for molecular weight distribution of high polymers". J Polym Sci A. ii (2): 835–843. doi:10.1002/pol.1964.100020220. ISSN 1542-6246.
- ^ Striegel A, Yau WW, Kirkland JJ, Bly DD (2009). Modernistic Size-Exclusion Liquid Chromatography: Exercise of Gel Permeation and Gel Filtration Chromatography (2nd ed.). Hoboken, North.J.: Wiley. ISBN9780470442876. OCLC 587401945.
- ^ Grubisic Z, Rempp P, Benoit H (1967). "A universal calibration for gel permeation chromatography". J Polym Sci B. 5 (nine): 753–759. Bibcode:1967JPoSL...5..753G. doi:10.1002/pol.1967.110050903. ISSN 1542-6254.
- ^ Sun T, Chance RR, Graessley WW, Lohse DJ (2004). "A Written report of the Separation Principle in Size Exclusion Chromatography". Macromolecules. 37 (11): 4304–4312. Bibcode:2004MaMol..37.4304S. doi:x.1021/ma030586k. ISSN 0024-9297.
- ^ Wang Y, Teraoka I, Hansen FY, et al. (2010). "A Theoretical Study of the Separation Principle in Size Exclusion Chromatography". Macromolecules. 43 (3): 1651–1659. Bibcode:2010MaMol..43.1651W. doi:10.1021/ma902377g. ISSN 0024-9297.
- ^ Kumar, Pranav (2018). Fundamentals and techniques of Biophysics and Molecular biology. New Delhi: Pathfinder Publication. p. 05. ISBN978-93-80473-15-iv.
- ^ "Size Exclusion Chromatograhy". pslc.ws. Polymer Science Learning Center (PSLC). 2005. Retrieved three January 2019.
- ^ Wyatt, Philip J. (1 Feb 1993). "Light scattering and the absolute label of macromolecules". Analytica Chimica Acta. 272 (1): 1–40. doi:10.1016/0003-2670(93)80373-South.
- ^ Podzimek, Stepan (April 5, 2014). "Truths and myths about the determination of molar mass distribution of synthetic and natural polymers by size exclusion chromatography". Journal of Practical Polymer Science. 131 (7): 40111. doi:x.1002/app.40111.
- ^ Some, Daniel; Amartely, Hadar; Tsadok, Ayala; Lebendiker, Mario (June 20, 2019). "Characterization of Proteins past Size-Exclusion Chromatography Coupled to Multi-Angle Light Handful (SEC-MALS)". Journal of Visualized Experiments (148): e59615. doi:ten.3791/59615. PMID 31282880. Retrieved October x, 2020.
- ^ Herold KE, Rasooly A (2009). Lab on a chip technology: Biomolecular separation and assay. Vol. two. Norfolk, UK: Horizon Scientific Press. p. 170. ISBN9781904455462. OCLC 430080586.
External links [edit]
Principle Of Size Exclusion Chromatography,
Source: https://en.wikipedia.org/wiki/Size-exclusion_chromatography
Posted by: pennywilsock.blogspot.com



0 Response to "Principle Of Size Exclusion Chromatography"
Post a Comment